List of Chapters
Impact of organophosphates and other pesticides on blood parameters of fishes and earthworms
Author(s): Singh S; Tiwari RK; Ghosh S; Pandey RS*
The earthworms and fishes are regarded as most agro-economically valuable organisms in India and around the world. The earth worms living underground can make the soil fertile by two means. First, it can flip the down soil upwards and secondly it can make the soil rich with nitrogen produced from waste products. In India and in other parts of the world, both fresh water and marine fishes are consumed largely. An indiscriminate use of pesticides in agriculture may stall the earthworm population and production of fishes by creating different physiological anomalies either by themselves or by their derivatives, though both of the organisms are non-target to the pesticides. Blood performs almost all important and vital physiological functions due to its circulatory nature. It transports necessary nutrients, gases and waste products throughout the body. Therefore, the present study was designed to explore the impact of various pesticides in modulations of hematological/cellular/metabolic and free radical parameters in blood of earthworms and fishes. We observed significant variations in different hematological parameters of these two organisms upon acute exposure to few organophosphates and other pesticides. These results suggest that the acute exposure of these pesticides can affect the non-target organisms and change the balance of eco-system adversely.
An introduction to blood groups and blood transfusion in domestic animals
Author(s): Aasif Ahmad Sheikh*; Showkat Ahmad Bhat; Ankita Kalyan; Mohammad Rayees Dar
Blood groups are decided by genetically influenced, polymorphic, antigenic components of the erythrocyte membrane. Blood group systems in general are independent of each other, and their inheritance conforms to Mendelian dominance. For polymorphic blood groups, an animal usually inherits 1 allele from each parent and thus expresses not more than 2 blood group antigens of a system. An exception being cattle, where multiple alleles or phenogroups are inherited. Normally, an individual does not have antibodies against any of the antigens present on its own red blood cells (RBC) or against other blood groups antigens of that species systems unless they have been induced by transfusion, pregnancy or immunization. The so called naturally occurring isoantibodies in some species (sheep, cattle, horses, pigs, dogs and cats), not induced by transfusion or pregnancy, may be present in variable but detectable titres. For example, cats with Group B essentially have anti-A antibody. With random blood transfusions in dogs, there is a 30-40% chance of sensitization of the recipient, primarily to blood group antigen DEA 1 (DEA stands for Dog Erythrocyte Antigen). ~50% of dogs have a naturally occurring cold hemagglutinin, DEA 7. In horses, transplacental immunization of the mare by an incompatible fetal antigen inherited from the sire may occur. As reported by Susan M. Cotter [1], immunization also may result when some homologous blood products are used as vaccines (e.g., anaplasmosis in cattle).
Inflammation and Platelet Aggregation: The Role of Blood Extracellular Purines and Adenosine
Author(s): Andreia Machado Cardoso*; Leandro Henrique Manfredi
Blood extracellular purines and adenosine, components of the purinergic system, are increasingly recognized as key mediators of activation and/or inhibition of circulating immune cells and platelets adhesion. This system is also composed by enzymes called ectonucleotidases and several receptors. In 1972, Burnstock introduced the concept of purinergic signaling. After that, the role of purines and adenosine as signalling molecules has been studied in virtually all organic systems. Nucleotides- particularly ATP- are well known for their function as a universal energy currency. Interestingly, ATP has a completely different role in the extracellular compartment, where it binds to nucleotide receptors and initiate a signaling cascade. These receptors are called purinergic P2 receptors. In contrast to P1 receptors, which are activated by adenosine, P2 receptors are activated by ATP and/or other nucleotides (e.g.: ADP and UDP).
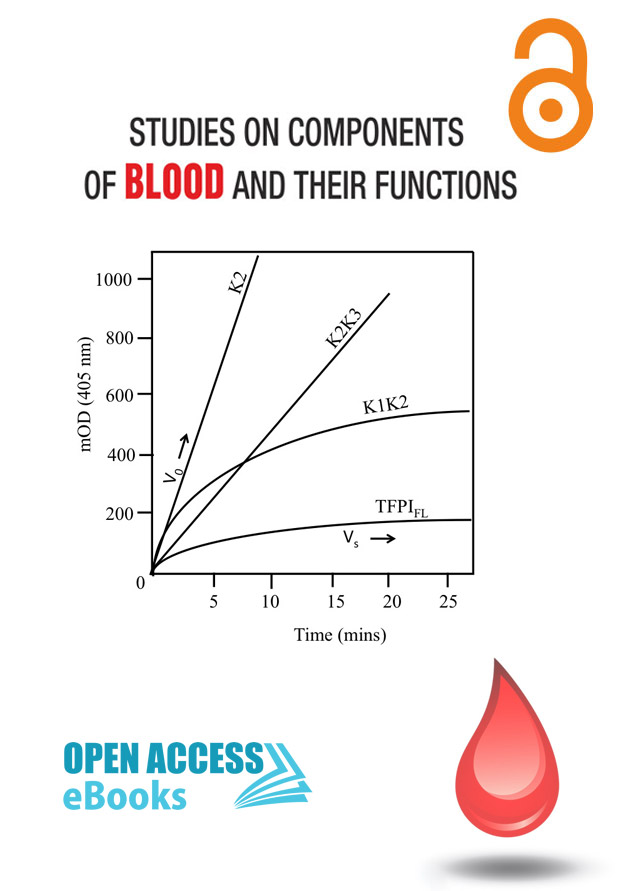
TFPI (Tissue Factor Pathway Inhibitor)
Author(s): Sameera Peraramelli
Blood coagulation is initiated as a response to vascular damage. Vascular injury exposes the integral membrane protein tissue factor (TF) [1,2] which binds the circulating coagulation factor VIIa (FVIIa) [3] and forms the TF-FVIIa complex. This complex activates FX [4,5] to FXa, which subsequently assembles with its cofactor factor Va (FVa) in presence of calcium ions and an appropriate phospholipid surface into the so-called prothrombinase complex.
Bacterial Bloodstream Infections
Author(s): Eric S Donkor*; Fleischer CN Kotey
Bacterial bloodstream infections have received increased attention over the years, and have seen a continuous expansion of the implicated etiologic agents. Of principal concern is the escalating rates of antimicrobial resistance observed in these etiologic agents. In this paper, the authors clarify major terminologies used in connection with bloodstream infections, review bloodstream infections in the context of bacterial etiologic agents and their associated antimicrobial resistance, and present a brief overview of the pathogenesis of bacterial bloodstream infections.
Coagulation Factors: An Overview
Author(s): Prajay K Rathore; Shaunak Mangeshkar; Huzefa Bhopalwala; Nakeya Dewaswala; Vishal Gaurav; Nishant R Tiwari*
Hemostasis is a critical physiological function defined as the process that leads to the cessation of bleeding from a blood vessel. Broadly, the process of hemostasis involves the following: local vasoconstriction, platelet plug formation, and the coagulation cascade. The initial brief reflexive vasoconstriction with platelet adhesion, aggregation, and activation constitutes primary hemostasis, while fibrin clot formation and stabilization are referred to as secondary hemostasis